Harmony Biosciences Holdings (HRMY)·Q4 2025 Earnings Summary
Harmony Biosciences Q4 2025 Earnings: Revenue Beats, But Patent Fears Trigger 25% Stock Plunge
February 24, 2026 · by Fintool AI Agent

Harmony Biosciences (NASDAQ: HRMY) reported Q4 2025 results that beat revenue expectations, but the story overshadowing earnings is a patent litigation loss that threatens the company's WAKIX franchise. The stock has cratered 25% over the past five days as investors digest the possibility of generic competition arriving years earlier than expected.
Key developments this quarter:
- Pediatric cataplexy FDA approval on February 13th, enabling full promotional efforts
- ANDA settlements completed with 6 of 7 generic filers, setting March 2030 generic launch (if pediatric exclusivity granted)
- Patient growth to ~8,500 average patients on WAKIX, with +400 patient additions in Q4 (third consecutive quarter of 400+ adds)
- Sales force expansion of ~20% announced across field sales, reimbursement, and remote teams
Did Harmony Biosciences Beat Earnings?
Revenue beat modestly, but the EPS picture is complicated by one-time charges.
Full Year 2025:
*Values retrieved from S&P Global
The Q4 profit decline reflects elevated R&D spending (+44% YoY to $49.9M) and G&A expenses (+99% YoY to $57.5M), the latter including milestone payments for pipeline assets.
What Did Management Guide?
Harmony guided 2026 net revenue of $1.00B-$1.04B, roughly in-line with analyst consensus of $1.009B.*
*Values retrieved from S&P Global
Management emphasized the company is "on track to achieve $1B+ in narcolepsy alone," with ~8,500 average patients on WAKIX and 80,000 diagnosed narcolepsy patients representing a large remaining opportunity. Pediatric patients represent ~5% of the narcolepsy population.
Q1 2026 Seasonality Note: CFO Sandeep Kapadia cautioned that Q1 typically sees "higher gross to net deductions due to insurance plans resets and higher copay obligations, along with potential for drawdown in trade inventories."
How Did the Stock React?
HRMY is down ~25% in 5 days—but this isn't about earnings.
The real catalyst for the stock collapse was a patent bench trial that concluded Friday, February 20, 2026 in Delaware District Court. AET Pharma, a generic manufacturer, is challenging Harmony's intellectual property on WAKIX. Reports indicate the judge "leaned negatively" toward Harmony during proceedings.
Analyst Reactions:
- Deutsche Bank: Downgraded to Hold from Buy, PT cut to $31 from $47
- Truist: Downgraded, citing increased uncertainty regarding WAKIX market exclusivity
If Harmony loses the case, WAKIX loss of exclusivity could occur as early as 2026 versus the previously expected 2030.
What Changed From Last Quarter?
Key operating metric changes:
The company continues to build cash, generating $348.2 million from operations in 2025 and ending Q4 with $882.5M in cash, cash equivalents, and investments—up $104M sequentially.
Expenses rose sharply due to pipeline investment and litigation:
- R&D up 44% YoY to $49.9M, reflecting Phase 3 trials for pitolisant HD and EPX-100
- G&A up 99% YoY to $57.5M, including ANDA litigation and settlement expenses
Sales Force Expansion: Management announced a ~20% increase in total field-based personnel: field sales +10%, field reimbursement +50%, remote sales +10%. Hires expected to be in place by end of Q1 2026.
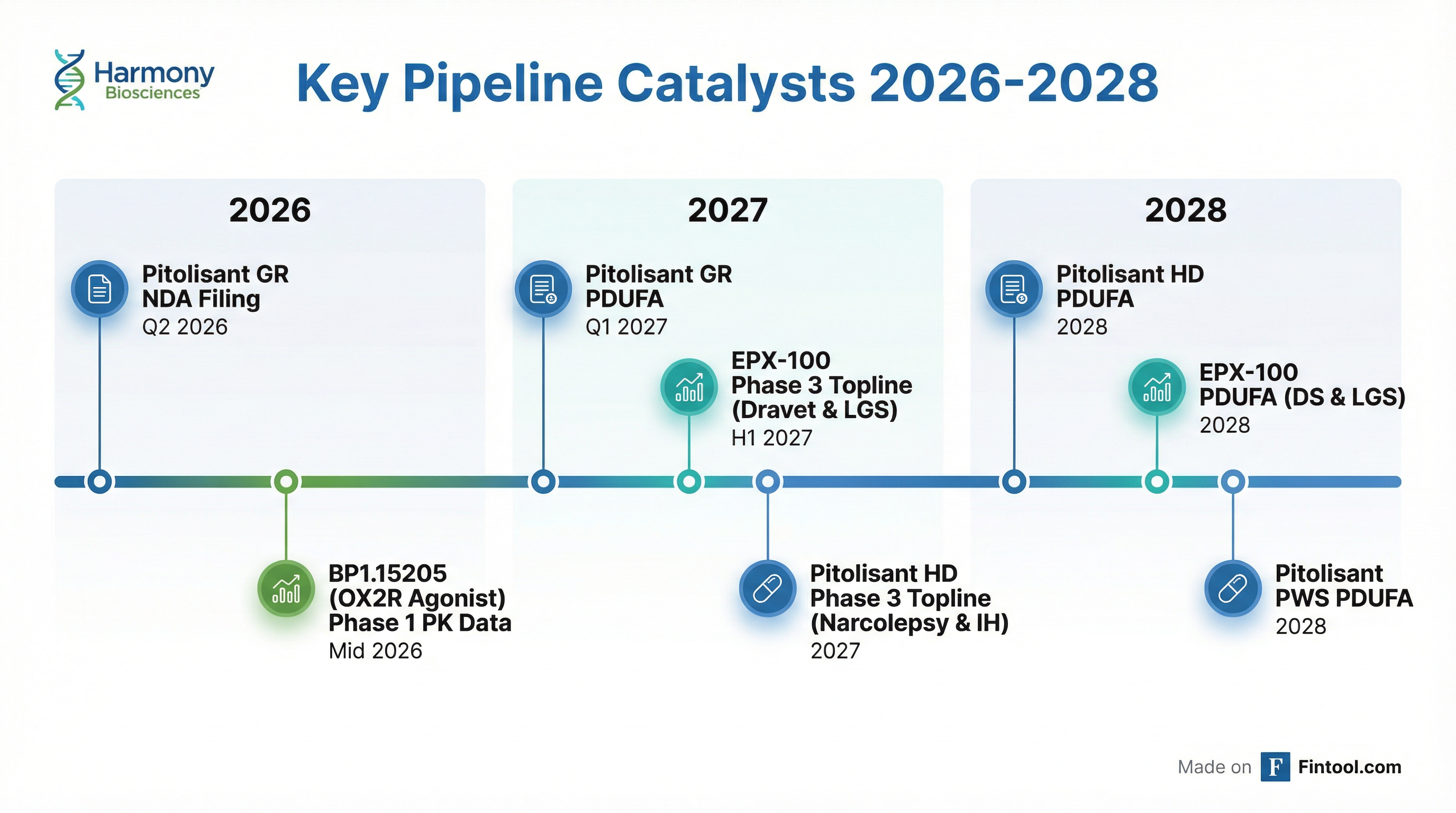
What's in the Pipeline?
Harmony outlined multiple catalysts through 2028, with 5 ongoing Phase 3 registrational trials across 5 distinct CNS indications and a potential 6th Phase 3 starting later this year:
Pitolisant Franchise Expansion:
- Pitolisant GR (Gastro-Resistant): NDA submission Q2 2026, PDUFA Q1 2027. Key differentiation: allows patients to start at therapeutic dose (17.8mg), eliminating the need for titration
- Pitolisant HD (High-Dose): Two Phase 3 trials ongoing—ONSTRIDE 1 (narcolepsy) and ONSTRIDE 2 (idiopathic hypersomnia). Both are 8-week, placebo-controlled trials with ~200 patients each. Pursuing differentiated labels: fatigue in narcolepsy and sleep inertia in IH. Topline data 2027, PDUFA 2028
- Pitolisant in PWS (TEMPO study): Phase 3 topline data anticipated 2H 2026; supports pediatric exclusivity extension. ~15,000 PWS patients in US, with ~half experiencing significant EDS
- NEW: MS Fatigue Exploration: Lead indication is fatigue in multiple sclerosis (~80% of MS patients have fatigue, >50% clinically significant). New formulation with issued patent until 2042. Currently focused on formulation optimization and exploring alternative modes of delivery
Novel Assets:
- BP1.15205 (OX2R Agonist): Phase 1 PK data expected mid-2026. Sleep-deprived healthy volunteer study planned for 2H 2026. Management is considering indications beyond narcolepsy/IH, including neuropsychiatric disorders involving cognition and mood
- EPX-100 (Clemizole): Phase 3 trials ongoing in Dravet Syndrome (ORCHEST study) and Lennox-Gastaut Syndrome (LIGHTHOUSE study); topline data H1 2027, PDUFA 2028. Open-label extension data presented at AES showed ~50% median seizure reduction with favorable tolerability—no significant GI AEs or liver function abnormalities, BID dosing
- EPX-200 (Liquid Lorcaserin): Pre-IND work ongoing for developmental and epileptic encephalopathies
Q&A Highlights
On EPX-100 Competitive Positioning (Pete Stavropoulos, Cantor Fitzgerald):
"The efficacy that we saw in this study...approximately 50% median reduction in seizures, and we also saw 50% reduction in seizures in about 50% of these patients. What's important is to see this alongside the safety and tolerability profile. We did not see significant nausea, vomiting, abdominal pain, diarrhea...Liver function tests also remained normal...A liquid formulation, BID dosing regimen is much more better suited in this patient population." — Dr. Kumar Budur, Chief Medical Officer
On Orexin-2 Development Strategy (David Amsellem, Piper Sandler):
"We are keeping an open eye and looking at everything, not just central disorders of hypersomnia, but other potential neuropsychiatric disorders, including several aspects of cognition, mood, and other things." — Dr. Kumar Budur
On Patent Litigation Timeline (Corinne Johnson, Goldman Sachs):
"In the meantime, there is a stay that's in place that extends to February of 2027. Based on the outcome of the trial, obviously an appeals process is available to both sides...we have settled with six of the seven ANDA filers." — Dr. Jeffrey Dayno, CEO
On Sales Force Expansion (Adam Zaeske, Chief Commercial Officer):
"We triggered an expansion of our field-based teams...Overall, it's about a 20% increase in total field-based personnel...field sales, more than 10%, field reimbursement, more than 50%, our remote sales teams, more than 10%. We expect those folks to be in place by the end of this quarter." — Adam Zaeske
On Cash and Business Development (Sandeep Kapadia, CFO):
"We're in a very strong position. We have over $880 million in cash on the balance sheet...we also have $150 million capacity on the share buyback." — Sandeep Kapadia
What Are the Key Risks?
-
Patent Litigation: The AET Pharma trial outcome could allow generic WAKIX as early as 2026 vs 2030 expected LOE. A stay is in place until February 2027, and appeals are available to both sides. Without pediatric exclusivity, settled generic entry would be September 2029; with it, March 2030. Regulatory exclusivity: EDS to March 2026, cataplexy to October 2027.
-
Single Product Concentration: WAKIX represents essentially all of Harmony's revenue; pipeline assets are years from commercialization
-
Pipeline Execution: Multiple Phase 3 readouts in 2027 carry binary event risk
-
Elevated Spending: OpEx growth outpacing revenue growth is compressing margins near-term
Forward Catalysts
Bottom Line
Harmony Biosciences delivered solid Q4 results with 21% revenue growth, added ~400 patients, and guided to $1B+ for 2026—but none of that matters right now. The stock is trading on the patent litigation outcome, which could dramatically shorten WAKIX's exclusivity window. Notably, Harmony has settled with 6 of 7 ANDA filers at a March 2030 generic launch date (contingent on pediatric exclusivity), but the AET Pharma case remains the wildcard. With the judge reportedly unfavorable to Harmony and analyst downgrades piling up, the stock has already priced in significant risk. Investors considering HRMY need to weigh the pipeline optionality (including the newly FDA-approved pediatric cataplexy indication) and $882M cash cushion against the very real possibility of generic competition arriving years ahead of schedule.